It is normal for iPSC morphology to change at different stages of a culture. 
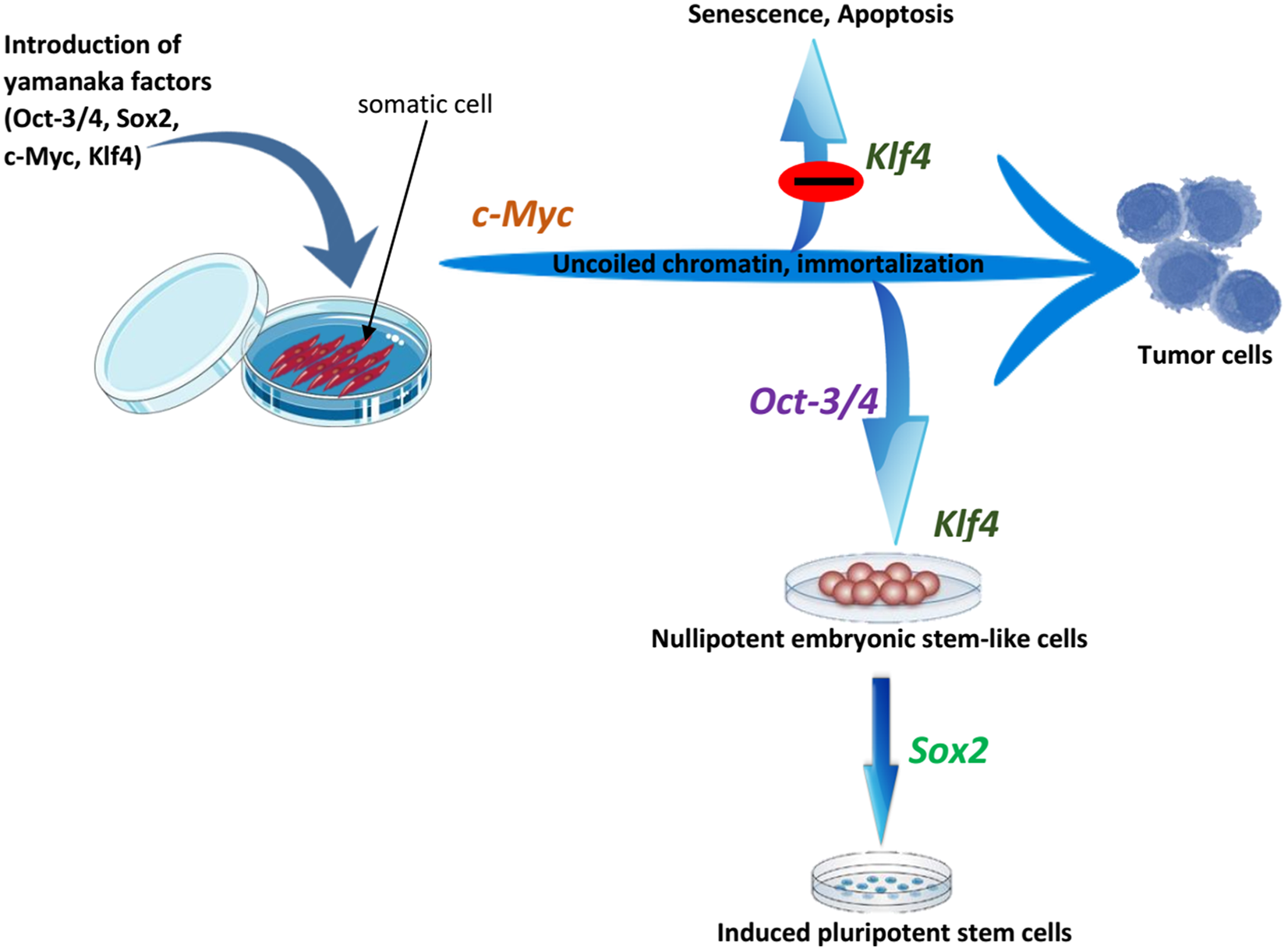
Observing the morphology of your iPSCs is the fastest and most effective way to monitor the quality of your iPSC cultures. What is important is that you find an approach that suits you, and more importantly, that you know how to properly evaluate the quality of your cultures. One lab using MEF feeders, mTESR medium, and colony passaging may have just as much success as another lab using feeder-free attachment substrates, Essential 8TM Medium, and single-cell passaging. Ultimately there is no one way to grow iPSCs. There are now more options than ever for reprogramming methods, growth media, cell attachment substrates, dissociation reagents, and passaging techniques. Achieving top-quality iPSC culturesĭespite all the advancements in iPSC technologies, culturing iPSCs still remains a challenging and complicated process. In the future, it might even be possible to grow entire organs from iPSCs that could be transplanted into humans10. This provides an unprecedented level of insight into diseases and an innovative platform for drug discovery. Examples include miniature kidneys, modeling the blood-brain barrier, and modeling the loss of nerve-muscle connectivity in motor neuron disease7-9. Developments in organoid and disease-on-chip technologies mean it is now possible to recreate tiny organ systems with different interacting cell types in a dish. Human cell types that were previously impossible to obtain by other means can now be derived from patient iPSCs with disease-specific genetic backgrounds. Disease modeling and drug discovery have now become more sophisticated than ever. Clinical trials have already begun in humans using iPSC-based stem cell therapies to treat age-related macular degeneration and Parkinson’s disease5,6. Since Yamanaka's breakthrough, the advances made using iPSCs have been unprecedented. These genes (C-Myc, SOX2, KLF4, and OCT4) are known as the ‘ Yamanaka Factors’, and the cells became known as induced pluripotent stem cells (iPSCs)4. Through a process of elimination, he found that just four of these genes, when over-expressed, were enough to rewire adult cells back into a pluripotent state. He made an educated guess of 24 genes that he thought might be able to do it, later describing this as like ‘buying a winning lottery ticket’. In the early 2000s, Shinya Yamanaka hypothesized against all conventional wisdom, stating that this wiring might be surprisingly simple to revert. The only reason they don’t, is that they are epigenetically hard-wired through the development to become a specialized cell type. Since the 1960s, thanks to pioneering work by John Gurdon, it has been known that all adult cells contain the complete genetic toolkit to make any other cell type. This provided an impetus to find another, less ethically controversial source of pluripotent stem cells. During this period federal funding for stem cell research in the USA was massively withdrawn3. These were termed ‘embryonic stem cells' (ESCs) and garnered a tremendous amount of controversy since discarded embryos from IVF were used to make them. Early work by Evans, Kaufman, and Thomson showed that pluripotent stem cells could be isolated from the inner cell mass of blastocyst-stage embryos and cultured indefinitely in vitro1,2. Researchers have long been conscious of the potential applications of pluripotent stem cells. At the other end of the spectrum, multipotent stem cells are found throughout development and adulthood, but can only produce a handful of different cell types. In contrast, totipotent stem cells share this ability, yet can also contribute to the extra-embryonic tissues, such as the placenta and umbilical cord. Pluripotent stem cells have the extraordinary capacity to become any cell type in the adult body, but unfortunately, only appear naturally for a very brief window during early embryonic development. The history and applications of iPSC research Just 6 years after publishing his work, Shinya Yamanaka went on to win the 2012 Nobel Prize in Physiology and Medicine and the burgeoning field of iPSC research has continued to flourish ever since. These so-called ‘induced pluripotent stem cells’ (iPSCs) had acquired the remarkable ability to become any cell type in the adult body.Ĭells lost to disease or injury could now theoretically be replaced using person’s own cells, and human cell types could be grown in the lab like never before. In 2006 Shinya Yamanaka published his ground-breaking research showing that adult cells could be re-wired into an embryonic-like state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
